As a CRO, your reputation depends on delivering compliant, submission-ready data, on time, every time. Sponsors demand quality, transparency, and regulatory confidence – and effective CRO clinical data management is essential in meeting these expectations.
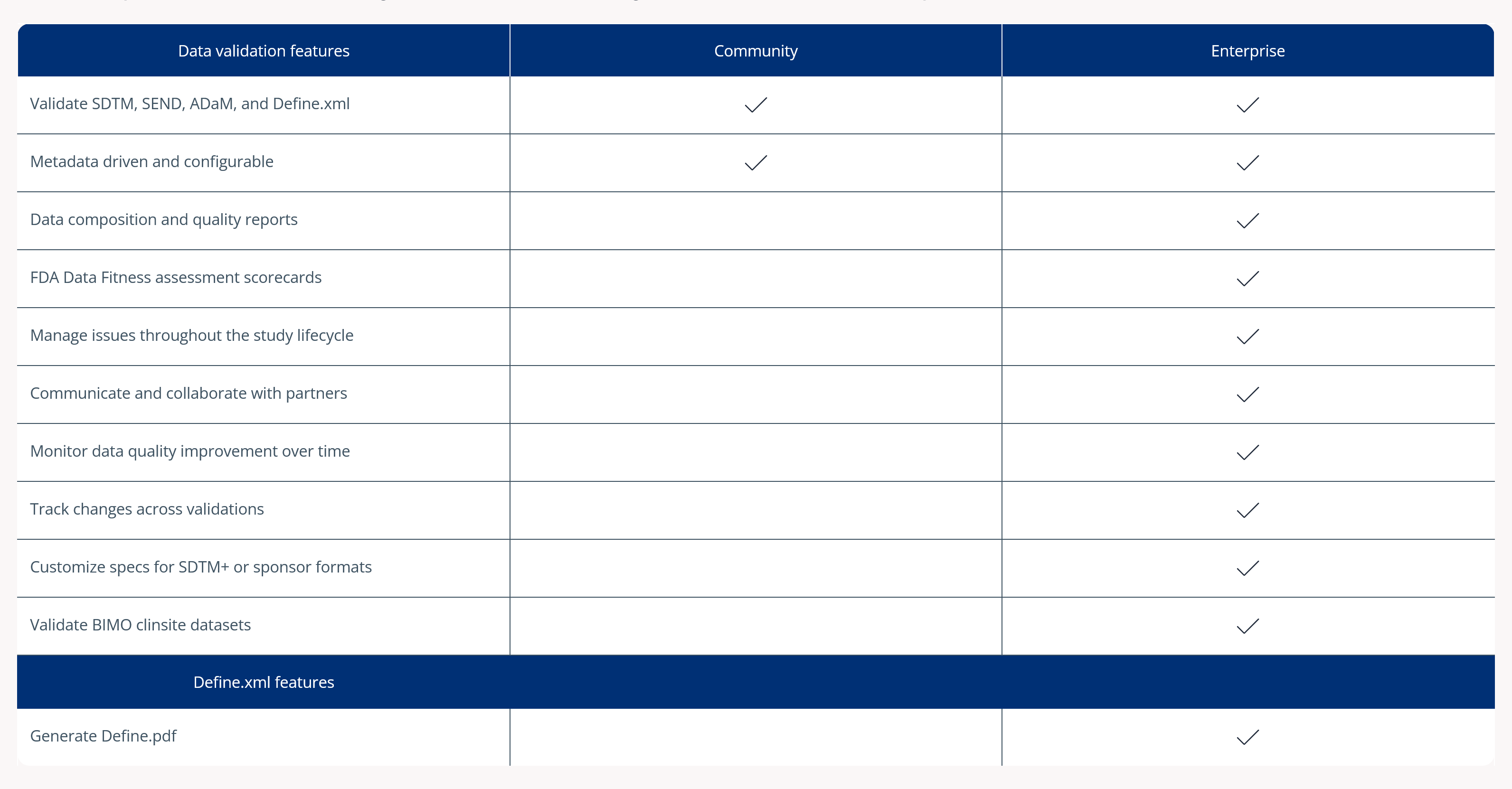
While Pinnacle 21 Community supports basic validation, it isn’t built for the scale, governance, and multi-study oversight CROs require. Managing multiple sponsors and global submissions goes beyond desktop tools and manual workflows.
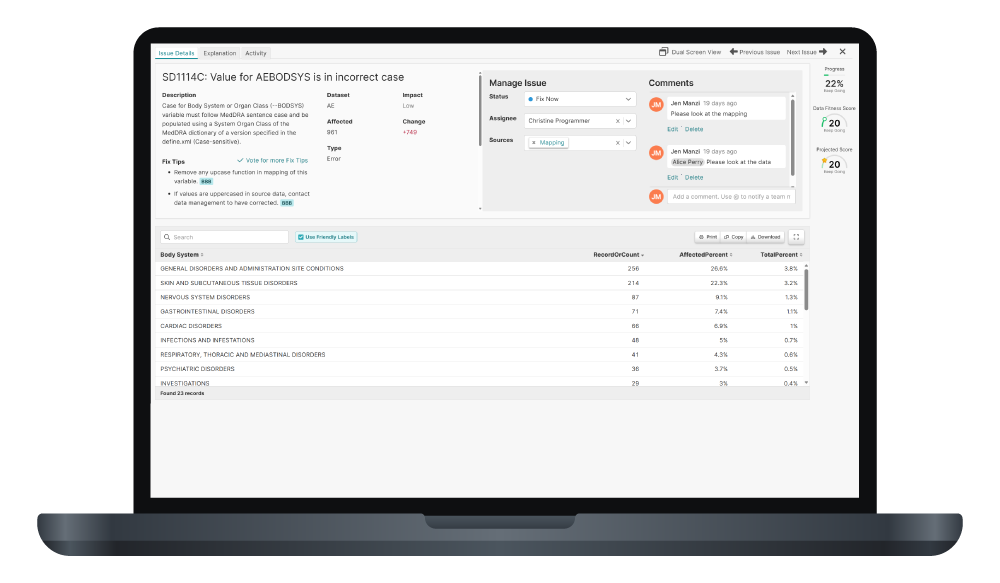
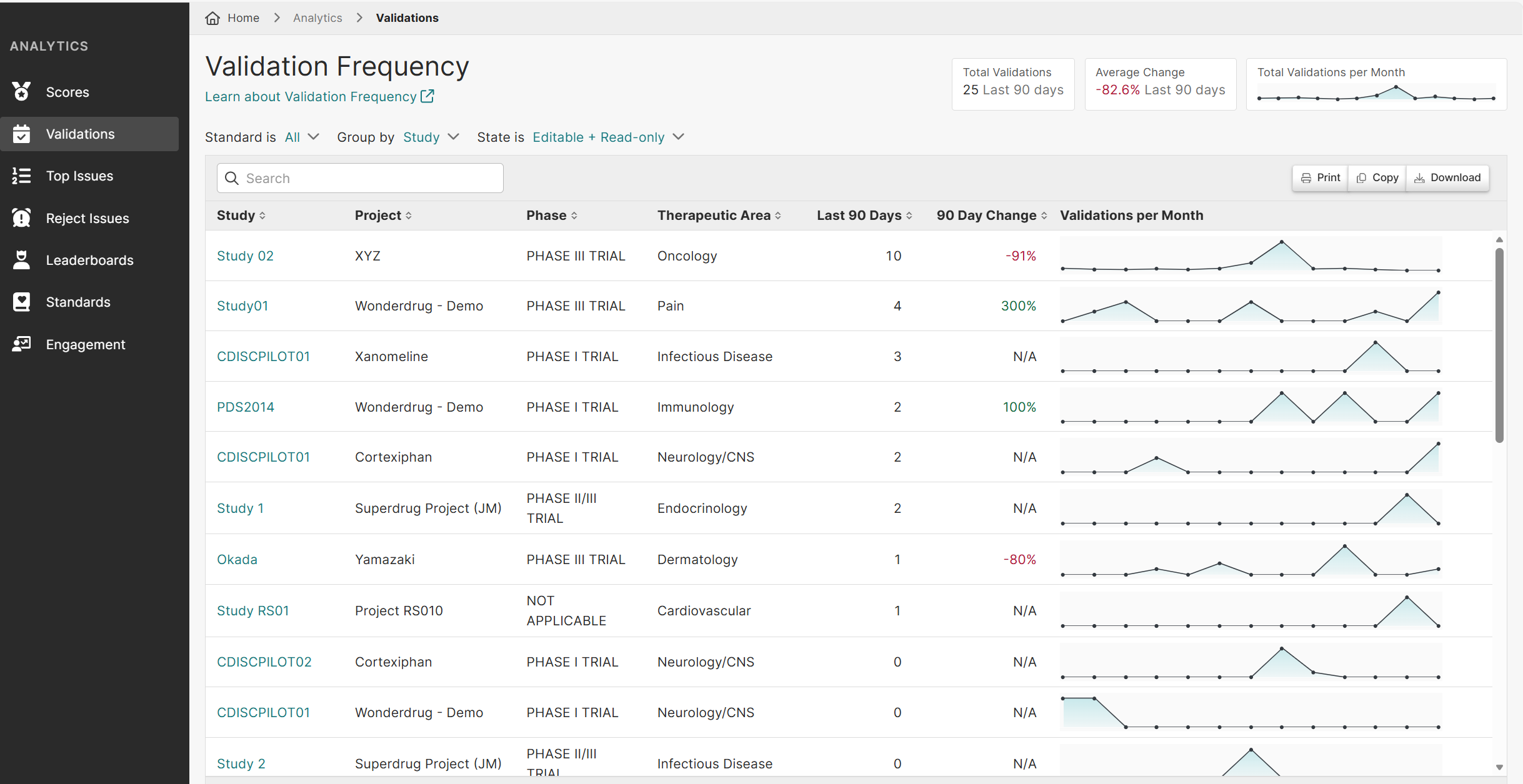
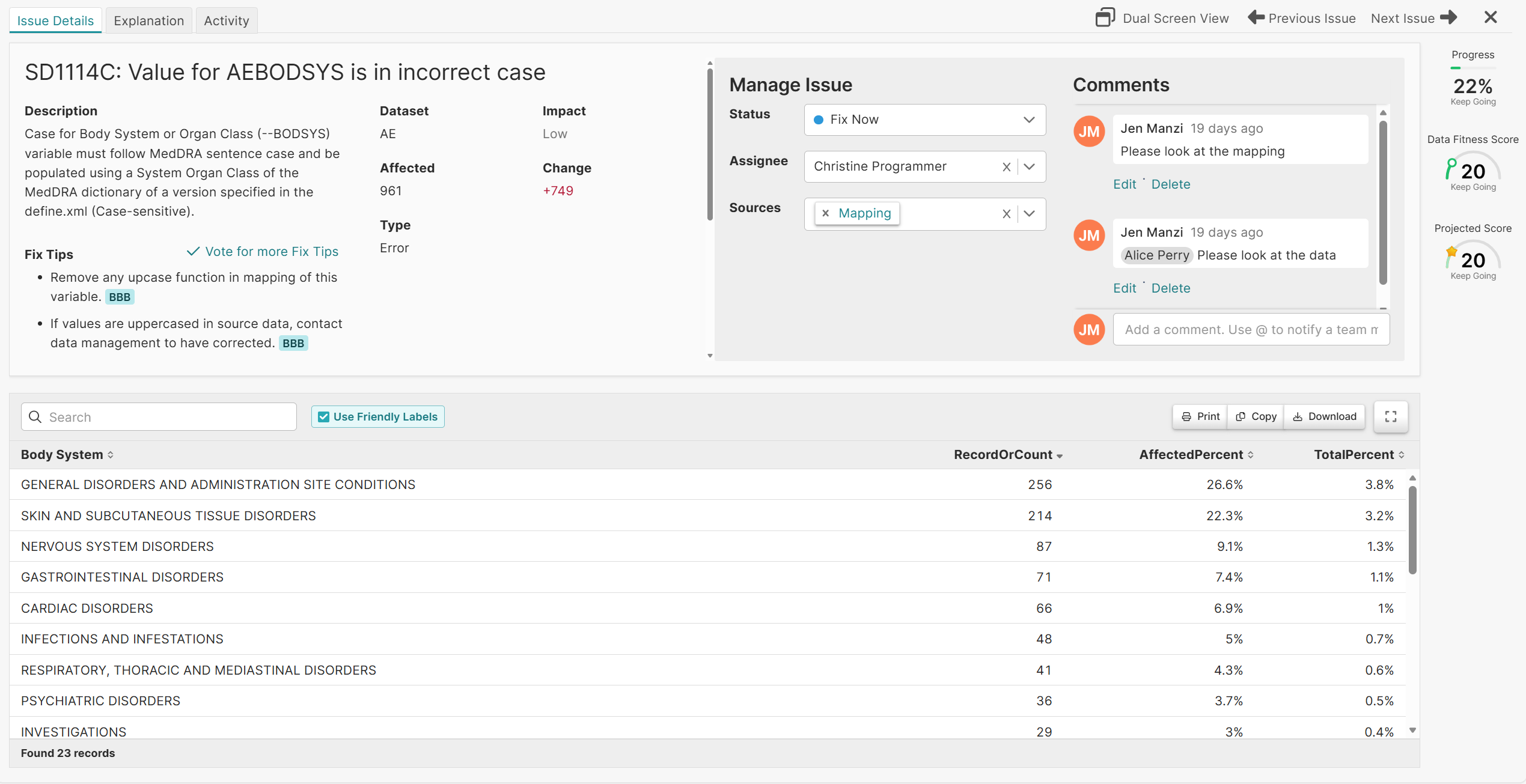
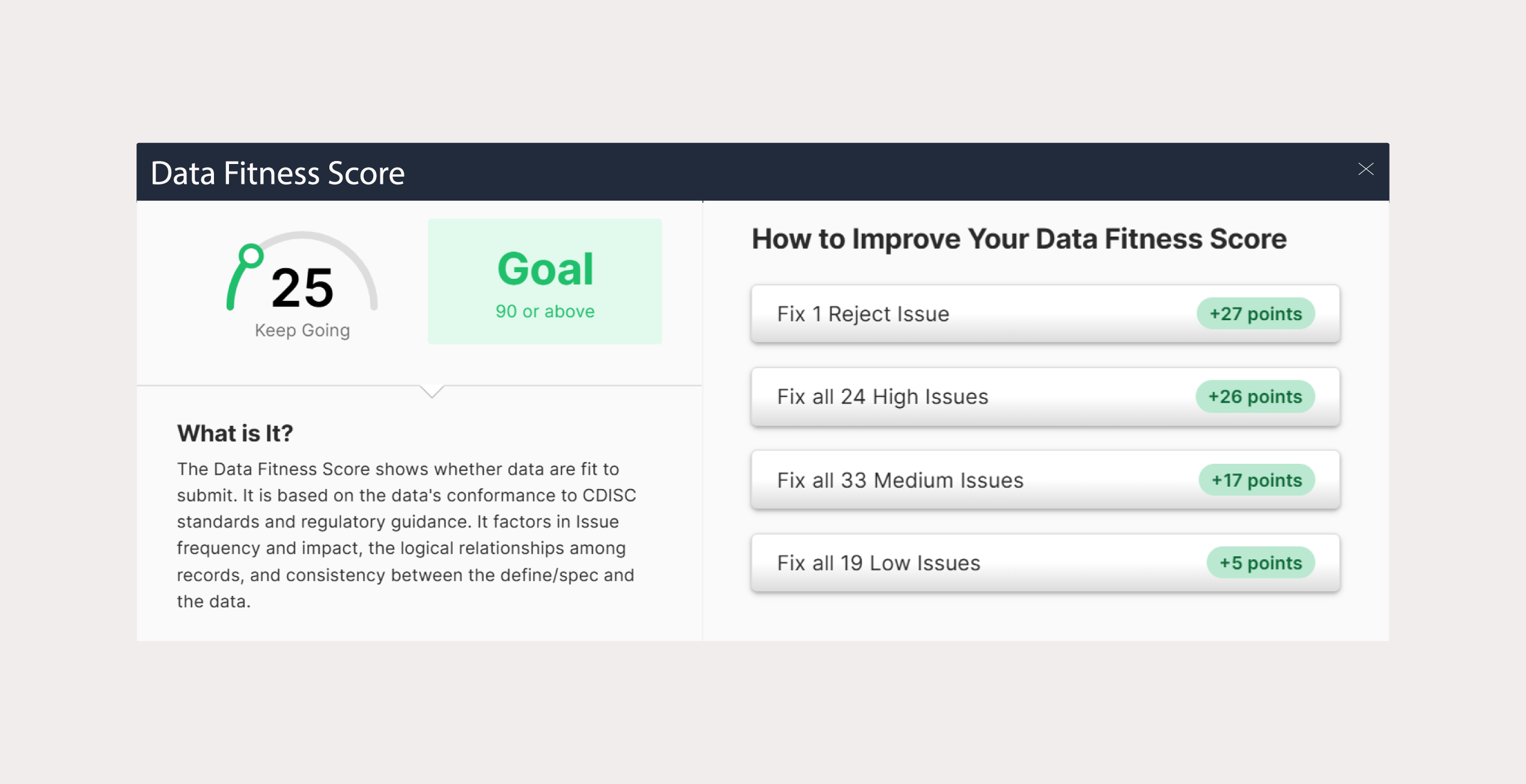
Pinnacle 21 Enterprise transforms validation into a centralized, enterprise-grade compliance framework. With Enterprise, CROs can confidently deliver regulatory compliant data faster – while increasing operational efficiency and strengthening competitive advantage.